AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Absolute entropy table4/9/2024 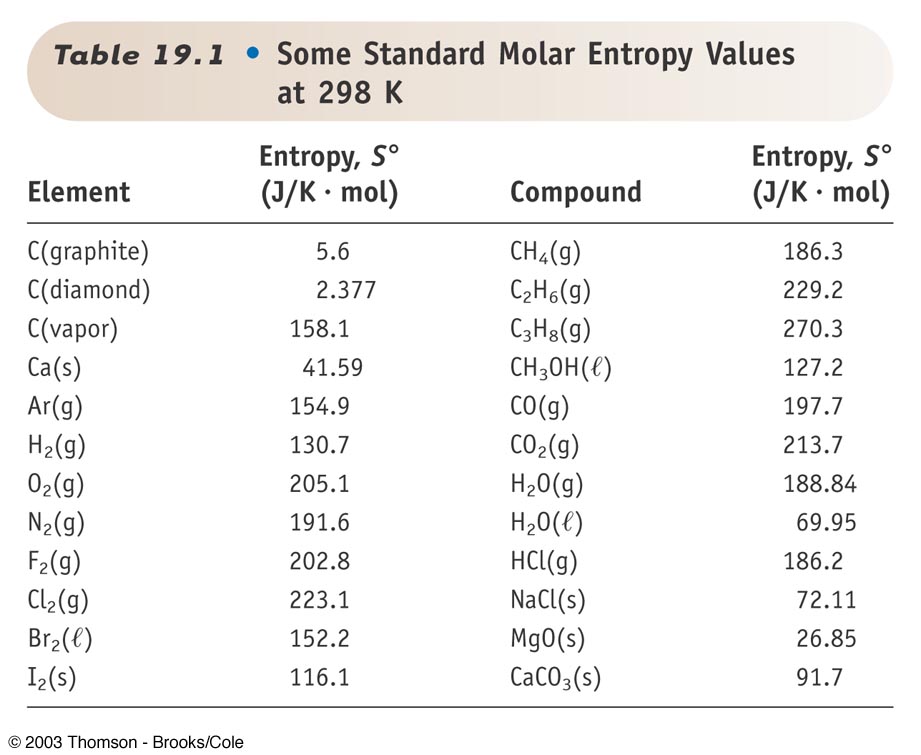
We write \(S^o_A\left(T\right)\) to indicate the absolute entropy of substance \(A\) in its standard state at temperature \(T\). It is usually included in compilations of thermodynamic data for chemical substances. The standard entropy is usually given the symbol \(S^o\). Thermodynamic databases contain information about thermodynamic properties for substances, the most important being enthalpy, entropy, and Gibbs free energy. When the entropy value is calculated for one mole of the substance in its standard state, the resulting absolute entropy is called the standard entropy. Where the substance undergoes phase changes, the contribution that the phase change makes to the entropy of the substance is equal to the enthalpy change for the phase change divided by the temperature at which it occurs.Īt any given temperature, the entropy value that is obtained in this way is called the substance’s absolute entropy or its third-law entropy. Free energy function referenced to 298.15 K. 
Phase changes are isothermal and reversible. Absolute entropy of a given substance, equal to zero at 0 K. In temperature ranges where experimental heat capacity data are available, the entropy change is obtained by integration using these data. \), using Debye’s theoretical relationship, \(C_P=AT^3\) \(A\) is obtained from the value of \(C_P\) at the lowest temperature for which an experimental value of \(C_P\) is available. Entropy can be calculated from a table of standard values just as enthalpy.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
